
About Xofigo®▼ (radium-223 dichloride) in mCRPC
For adult patients with metastatic castration-resistant prostate cancer (mCRPC)
Xofigo (+ best standard of care) is the first and only targeted alpha therapy to improve overall survival and slow down the decline of quality of life during on treatment period vs placebo + best standard of care (BSC) in mCRPC patients with symptomatic bone metastases and no known visceral metastases.
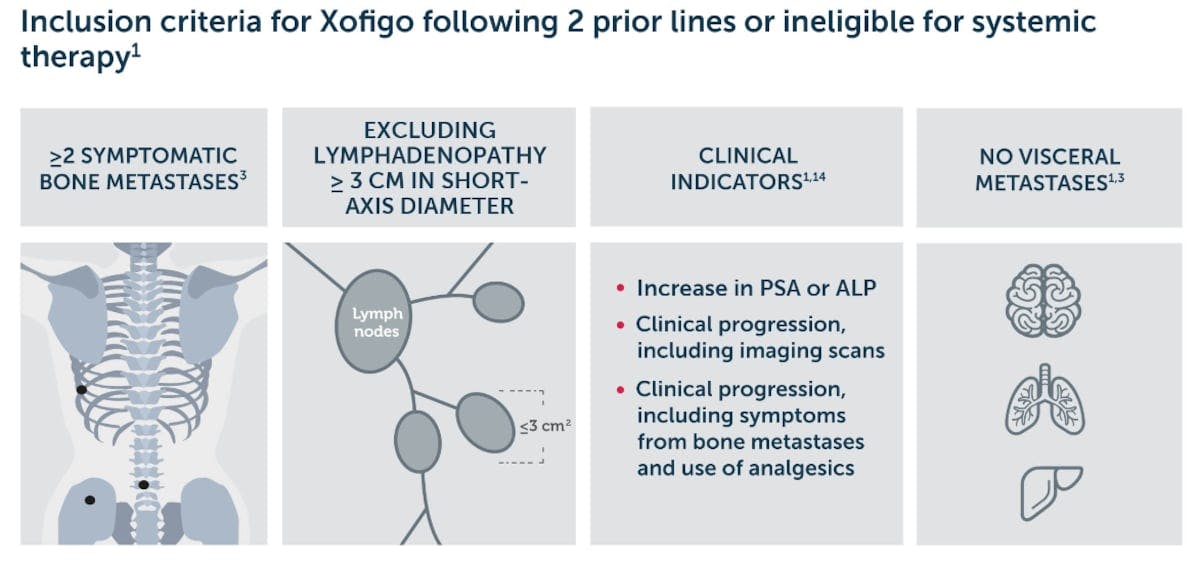
Xofigo monotherapy or in combination with luteinising hormone releasing hormone (LHRH) analogue is indicated for the treatment of adult patients with mCRPC, symptomatic bone metastases and no known visceral metastases, in progression after at least two prior lines of systemic therapy for mCRPC (other than LHRH analogues), or ineligible for any available systemic mCRPC treatment.
- Fully funded in the UKexpand_moreXofigo is fully funded throughout the UKApproved by NICE
Xofigo is NICE recommended as an option for treating patients with hormone-relapsed prostate cancer with 2 or more symptomatic bone metastases and no known visceral metastases if they have experienced disease progression after at least 2 lines of prior systemic therapy (other than LHRH analogues) or are ineligible for available systemic therapy options.
SMC acceptedFor the treatment of adults with CRPC, symptomatic bone metastases and no known visceral metastases
To find out more, see Guidelines
- Patient identification & Treatment sequencingexpand_moremCRPC patients with symptomatic bone metastases should be treated early with Xofigo (prior to visceral metastases)Introduce Xofigo for your mCRPC patients who have progressed on two lines of systemic therapy (other than LHRH analogues) or if ineligible for systemic therapy.Identifying the right patients for treatment with Xofigo
 How Xofigo may fit in to your treatment sequence
How Xofigo may fit in to your treatment sequence
These are illustrative algorithms based on the revised EU indication for Xofigo. Not all potential treatment options are shown. Concurrent use of bone health agents to treat osteoporosis or for patients with bone metastases is recommended. Sequential use of abiraterone and enzalutamide (or vice versa) is not considered as an option due to likely futility of treatment. Consider clinical trials or best supportive care after all available systemic therapies have been administered.
Abbreviations: ADT: androgen deprivation therapy; AR: androgen receptor; EU: European Union; LHRH: Luteinising hormone releasing hormone; mCRPC: metastatic castration-resistant prostate cancer; mHSPC: metastatic hormone-sensitive prostate cancer; nmCRPC: non-metastatic castration-resistant prostate cancer.
Use Xofigo while there is an opportunity for action before the development of visceral metastases - Efficacyexpand_moreXofigo + BSC significantly extended median OS vs placebo + BSC
In the phase 3 ALSYMPCA trial, an updated analysis showed that Xofigo + BSC significantly extended median OS by 3.6 month vs placebo + BSC (14.9 months vs 11.3 months, respectively; P<0.001).
Median OS
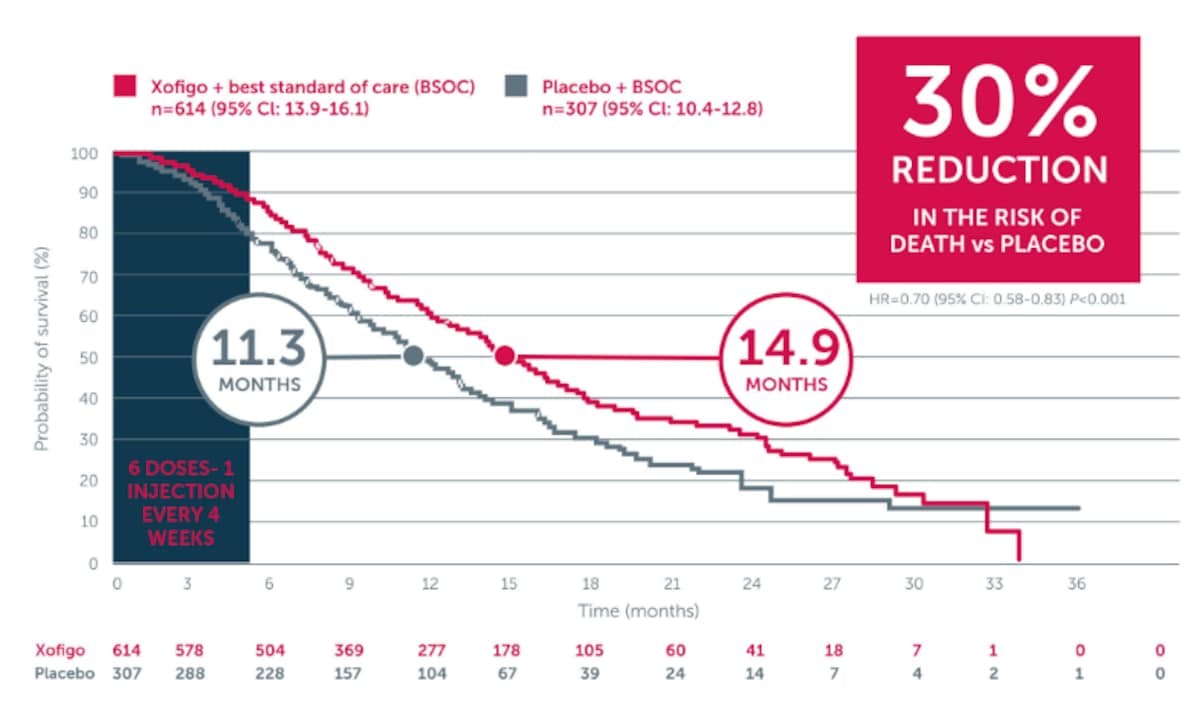
Adapted from Parker C et al. 2013. ALSYMPCA was a double-blind, randomised, placebo-controlled, phase 3 study in 921 symptomatic mCRPC patients with 2 or more bone metastases. Patients were randomised 2:1 to receive 6 injections (one every 4 weeks) of Xofigo + BSC (n=614) or placebo + BSC (307). BSC included local EBRT or treatment with bisphosphonates, corticosteroids, antiandrogens, oestrogens, estramustine, or ketoconazole. The primary endpoint was overall survival.
Abbreviations: BSC: best standard of care; EBRT: external beam radiation therapy; mCRPC: metastatic castration-resistant prostate cancer; OS: overall survival.
Xofigo + BSC significantly delays time to first symptomatic skeletal event (SSE) vs placebo + BSCIn the phase 3 ALSYMPCA trial, an updated analysis of secondary endpoints showed that Xofigo + BSC significantly delayed time to first SSE by 5.8 months vs placebo + BSC (15.6 months vs 9.8 months, respectively; P<0.001).
Median time to first SSE
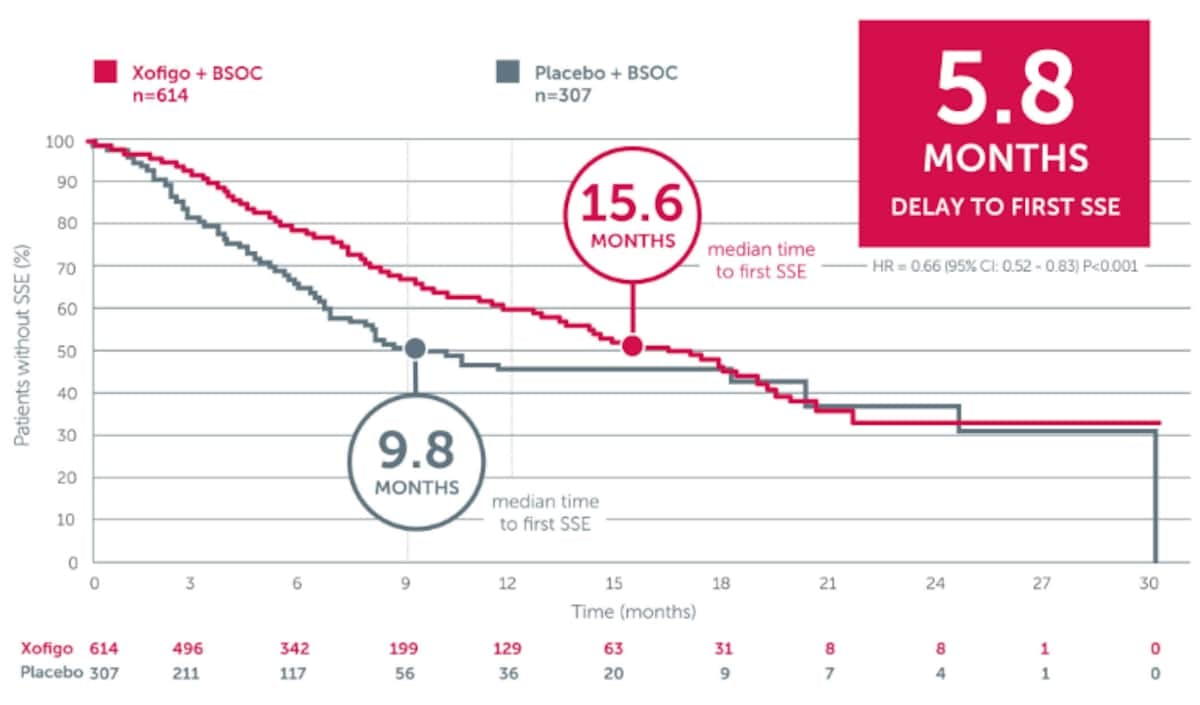
Adapted from Parker C et al. 2013. ALSYMPCA was a double-blind, randomised, placebo-controlled, phase 3 study in 921 symptomatic mCRPC patients with bone metastases. Patients were randomised 2:1 to receive 6 injections (one every 4 weeks) of Xofigo + BSC (n=614) or placebo + BSC (307). BSC included local EBRT or treatment with bisphosphonates, corticosteroids, antiandrogens, oestrogens, estramustine, or ketoconazole. SSEs were defined as EBRT for pain relief, spinal cord compression, tumour-related orthopaedic surgical interventions, bone fractures. The primary endpoint was overall survival.
Abbreviations: BSOC: best standard of care; EBRT: external beam radiation therapy; mCRPC: metastatic castration-resistant prostate cancer; OS: overall survival; SSE: symptomatic skeletal event.
Improved survival with Xofigo + BSC is accompanied by a slower decline in quality of life vs placebo + BSCA post hoc analysis of the phase 3 ALSYMPCA trial showed that significantly more patients treated with Xofigo + BSC experienced a meaningful improvement in quality of life (as measured by FACT-P total score or EQ-5D utility score) vs placebo + BSC (P=0.02 and P=0.004, respectively).
In addition, significantly more patients experienced pain relief with Xofigo + BSC than placebo + BSC (30.2% [129/427] vs 20.1% [37/184], respectively; P=0.01)
Patients with a meaningful improvement in FACT-P total score and EQ-5D utility score
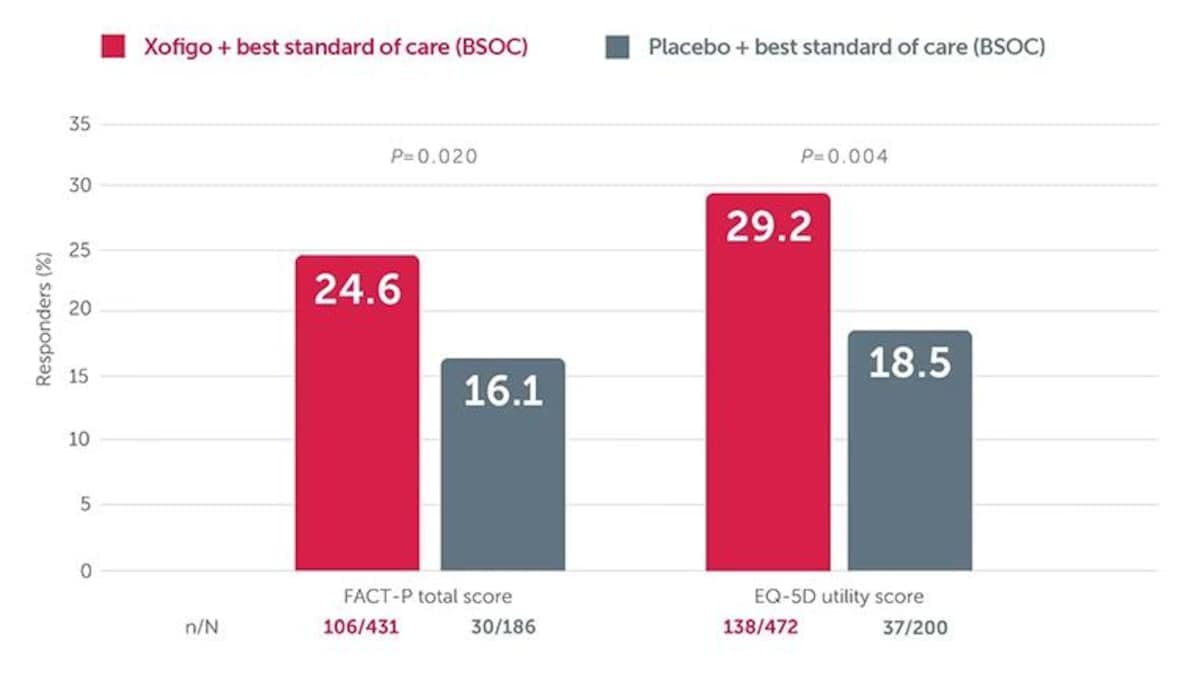
Adapted from Nilsson S et al. 2016. ALSYMPCA was a double-blind, randomised, placebo-controlled, phase 3 study in 921 symptomatic mCRPC patients with bone metastases. Patients were randomised 2:1 to receive 6 injections (one every 4 weeks) of Xofigo + BSC (n=614) or placebo + BSC (307). BSC included local EBRT or treatment with bisphosphonates, corticosteroids, antiandrogens, oestrogens, estramustine, or ketoconazole. The primary endpoint was overall survival. A post hoc analysis looked at health-related quality of life using two validated instruments: the general EQ-5D and the disease-specific FACT-P. Meaningful improvement defined as increase in FACT-P total score of ≥10 from baseline or an increase in EQ-5D utility score of ≥0.1 from baseline, at Week 16 and/or Week 24.
Abbreviations: BSOC: best standard of care; EBRT: external beam radiation therapy; EQ-5D: EuroQoL 5D; FACT-P: Functional Assessment of Cancer Therapy-Prostate; mCRPC: metastatic castration-resistant prostate cancer.
- Safetyexpand_moreSafety profile of Xofigo + BSC was comparable to placebo + BSC
In the phase 3 ALSYMPCA trial, the rates of AEs were similar with Xofigo + BSC vs placebo + BSC.
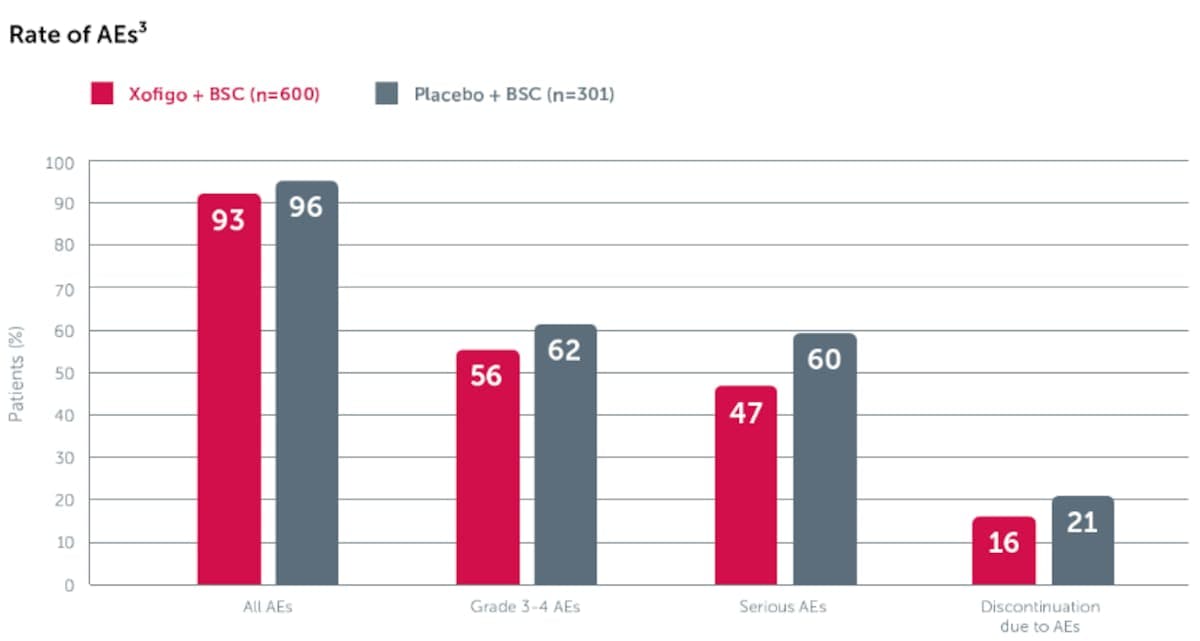
ALSYMPCA was a double-blind, randomised, placebo-controlled, phase 3 study in 921 symptomatic mCRPC patients with bone metastases. The safety population included 600 patients in the Xofigo + BSC group and 301 patients in the placebo + BSC group.
- The most frequently observed adverse reactions (≥ 10%) in patients receiving Xofigo were diarrhoea, nausea, vomiting, thrombocytopenia and bone fracture
- The most serious adverse reactions were thrombocytopenia and neutropenia
- Xofigo increases the risk of bone fractures. In clinical studies, concurrent use of bisphosphonates or denosumab reduced the incidence of fractures in patients treated with Xofigo
For the full list of AEs, please refer to the SMPC links below:
Treatment considerations- Chemotherapy may be used after Xofigo*
*The haematological safety profiles for patients receiving chemotherapy after Xofigo were similar to those seen in patients receiving chemotherapy after placebo.
Patient considerations- Low incidence of Grade 3–4 vomiting
- Can be administered without the need for steroids†
†Xofigo is contraindicated in combination with abiraterone acetate and prednisone/prednisolone. Concomitant use of steroids may further increase the risk of fracture.
Abbreviations: AE: adverse event; BSOC: best standard of care; mCRPC: metastatic castration-resistant prostate cancer.
- Dosingexpand_moreXofigo has a fixed course of treatmentXofigo is administered as a 1-minute IV injection every 4 weeks for 6 injections.
 Help your patients experience the benefits of Xofigo
Help your patients experience the benefits of Xofigo
Use of Xofigo is not recommended in patients with low levels of osteoblastic bone metastases.
Abbreviations: OS: overall survival; SSE: symptomatic skeletal event; QoL: quality of life
PP-XOF-GB-0460 | February 2024
- Referencesexpand_more
Adverse events should be reported. Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/ or search MHRA Yellow Card in Google Play or Apple App Store. Adverse events should also be reported to Bayer plc.
If you want to report an adverse event or quality complaint, reports can be directed to Tel: 0118 206 3500 or Email: pvuk@bayer.com.
Further information is available on the "contact" tab at www.bayer.co.uk.
Register with BayerPRO
To access additional resources, personalise your experience and keep up to date via electronic communications from Bayer.
Prescribing Information (PI) can be found via the links below:
Xofigo (radium-223 dichloride)
Bayer Pro is owned and funded by Bayer plc
PP-PF-ONC-GB-0396 | February 2024